-
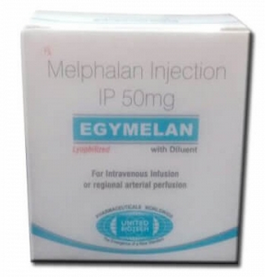
EGYMELAN 50 mg Melphalan Injection
WHAT IS EGYMELAN?
EGYMELAN 50mg is one of a group of drugs called alkylating agents. It
sticks to one of the cancer cell’s DNA strands. DNA is the genetic code that is
in the nucleus of all animal and plant cells. It controls everything the cell
does. The cell cannot then grow and divide into 2 new cells.
MEDICINE USES :
Melphalan is used to treat multiple myeloma (a type of blood cancer),
advanced breast cancer and cancer of the ovary. This medicine treats only the
symptoms of ovarian cancer or multiple myeloma, but does not treat cancer
itself.
MELPHALAN COMMON SIDE
EFFECTS :missed menstrual periods; weakness; or temporary hair loss.
Call your doctor at once if you have:
bone marrow suppression–sudden weakness or ill feeling, fever, chills,
pale skin, easy bruising or bleeding, red or pink urine, painful mouth sores,
cough, trouble breathing, feeling light-headed, rapid heart rate;
inflammation of your blood vessels–numbness or tingling, red skin rash,
unusual lumps or masses, fever, weight loss, muscle or joint pain, tired
feeling, unusual bleeding; or
liver problems – nausea, upper stomach pain, itching, loss of appetite,
dark urine, clay-colored stools, jaundice (yellowing of the skin or eyes).
This is not a complete list of side effects and others may occur. Call
your doctor for medical advice about side effects.
SAFETY INFORMATION
(Precautions) for EGYMELAN MELPHALAN INJECTION :Egymelan may lower the ability of your body to fight infection. Avoid
contact with people who have colds or infections. Tell your doctor if you
notice signs of infection like fever, sore throat, rash, or chills.
Do not receive a live vaccine (eg, measles, mumps) while you are taking
Melphalan. Talk with your doctor before you receive any vaccine.
Use of Egymelan may increase your risk of developing another type of
cancer. The risk may be greater if you use higher doses of this medicine or if
you use it for a longer period of time. Discuss any questions or concerns with
your doctor.
Egymelan may affect the ovaries. This may cause irregular or absent
menstrual periods and decreased fertility in some women. Discuss any questions
or concerns with your doctor.
Egymelan may affect the testicles and cause decreased fertility in some
men. This may be permanent in some patients. Discuss any questions or concerns
with your doctor.
PREGNANCY and BREAST-FEEDING: Melphalan has been shown to cause harm to
the fetus. Avoid becoming pregnant while taking it. If you think you may be
pregnant, contact your doctor. You will need to discuss the benefits and risks
of using this medicine while you are pregnant. It is unknown if Generic
Melphalan is found in breast milk. Do not breast-feed while taking it.This is not a complete list. Ask your health care provider for more
information.
MEDICATION STORAGE
REQUIREMENTS :Store at 20° to 25°C (68° to 77°F).
DRUG INTERACTIONS :
Tell your health care provider if you are taking any other medicines,
especially any of the following:
Cisplatin because the risk of Melphalan’s side effects
may be increased
Carmustine (BCNU), cyclosporine, or nalidixic acid
because serious lung, kidney, or bowel problems may occur
Palifermin because if mouth or tongue sores develop,
they may be more severe or last longer
This may not be a complete list of all interactions that may occur. Ask
your health care provider if Melphalan may interact with other medicines that
you take. -
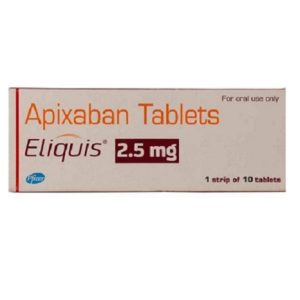
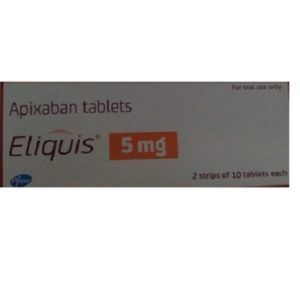
Elbonix 艾曲波帕 Tablets
产品介绍:
艾曲波帕由英国葛兰素史克公司研发并于2008年11月获得美国FDA批准在美国上市。艾曲波帕是一种促血小板生成素受体激动剂适用于治疗慢性免疫性血小板减少性紫癜患者的血小板减少,对皮质激素、免疫球蛋白或脾切除反应不佳的患者。艾曲波帕只应用于有ITP其血小板减少程度和临床情况增加出血风险的患者,不应用于意向正常血小板计数正常化。
目前,艾曲波帕已获全球100多个国家批准,用于慢性免疫(特发性)血小板减少性紫癜(ITP)患者血小板减少症的治疗,同时已获43个国家批准用于慢性丙型肝炎(CHC)患者血小板减少症的治疗,以便启动并维持以干扰素为基础的肝病标准疗法。
与现有治疗血小板减少症药物比较,艾曲波帕是可以口服给药的治疗血小板减少症的药物,安全性高。
碧康制药生产的Elbonix是该药在全球的首仿药,也是迄今为止唯一获得政府监管机构批准合法生产的仿制药。
碧康制药由欧洲财团参与投资,是南亚地区唯一执行欧盟技术规范,并且在孟加拉两大证券交易所主板上市的大型制药企业,产品符合欧洲药典和美国药典标准。
相比其他来源不清晰的所谓同一种仿制药,即使其主要成份含量与碧康产品相接近,但由于不具备严格规范的GMP标准生产场所和政府部门的严格监管,其产品中影响药物吸收率的溶出度和生物利用度也与碧康产品具有很大差异。
为保证用药安全,敬请谨慎选择!
适应症:
艾曲波帕是一种促血小板生成素受体激动剂适用于治疗慢性免疫性血小板减少性紫癜患者的血小板减少,对皮质激素、免疫球蛋白或脾切除反应不佳的患者。
艾曲波帕只应用于有ITP其血小板减少程度和临床情况增加出血风险的患者,不应用于意向正常血小板计数正常化。
艾曲波帕还可用于慢性丙型肝炎(CHC)患者血小板减少症的治疗,以便启动并维持以干扰素为基础的肝病标准疗法。
用法与用量:
艾曲波帕的起始剂量是50 mg每天1次;对东方人患者或中度或严重肝功能不全患者,起始剂量为25 mg每天1次,空胃给药(餐前1小时或2小时)。
艾曲波帕和其它药物、食物或多价阳离子(如,铁、钙、铝、镁、硒和锌)添加剂间允许间隔4小时。为减低出血风险调整每天剂量至达到和维持血小板计数≥50 X 109/L。每天剂量不要超过75 mg。
服用两周,以两周为周期加量至最大剂量每天150mg。主要终点是血液反应,12周之后进行初次评估。如果16周之后没有观察到血液反应就停止治疗。
-
WhatsApp / Viber
Available On All Major Platform
+91 7428091874
Express Worldwide Shipping
Order Above $99